 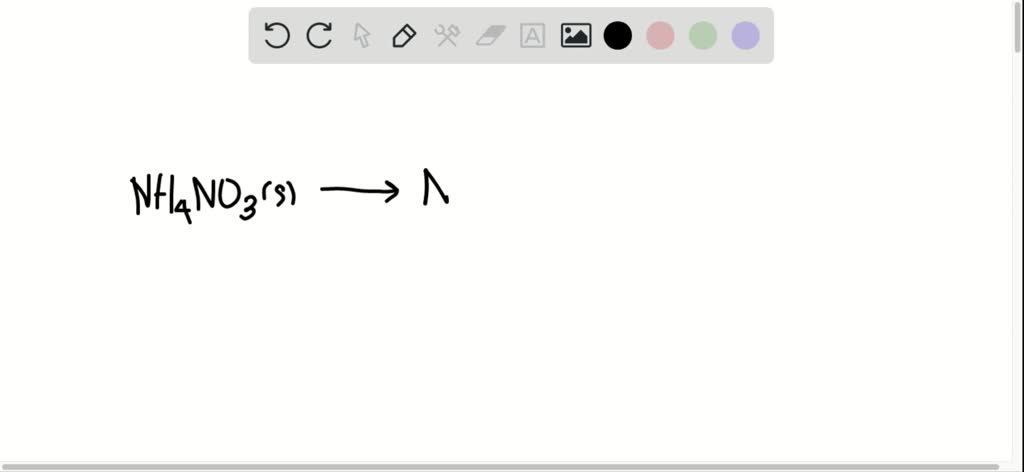
element by mass mass of element in compound molar mass of compound × 100 (6.9.1) (6.9.1) element by mass mass of element in compound molar mass of compound × 100. Find the mass of each of the products formed. The easiest way to do this is to calculate the molar mass of each compound and to use the portion of that mass contributed by nitrogen to calculate the percentage. NH4NO3(s) N2O(g) + 2H2O(l) NH 4 NO 3 ( s) N 2 O ( g) + 2 H 2 O ( l) In a certain experiment, 45.7g 45.7 g of ammonium nitrate is decomposed. Saliva and urine were analyzed for volatile N-nitrosamines, nitrate & nitrite.Blood was analyzed for nitrate. Ammonium nitrate decomposes to dinitrogen monoxide and water, according to the following equation. Samples of blood, saliva & urine were collected just before a 24 hr period. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. Add them together: add the results from step 3 to get the total molar mass of the compound. 12 Healthy volunteers ingested orally 7-10.5 g ammonium nitrate in a single dose & 12 others received 9.5 g of sodium nitrate iv in approx 60 min. Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. And don’t forget to put the unit g/mol to your final calculated molar mass. The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). 
First solve the brackets, then multiplications and at last do the final addition.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
