 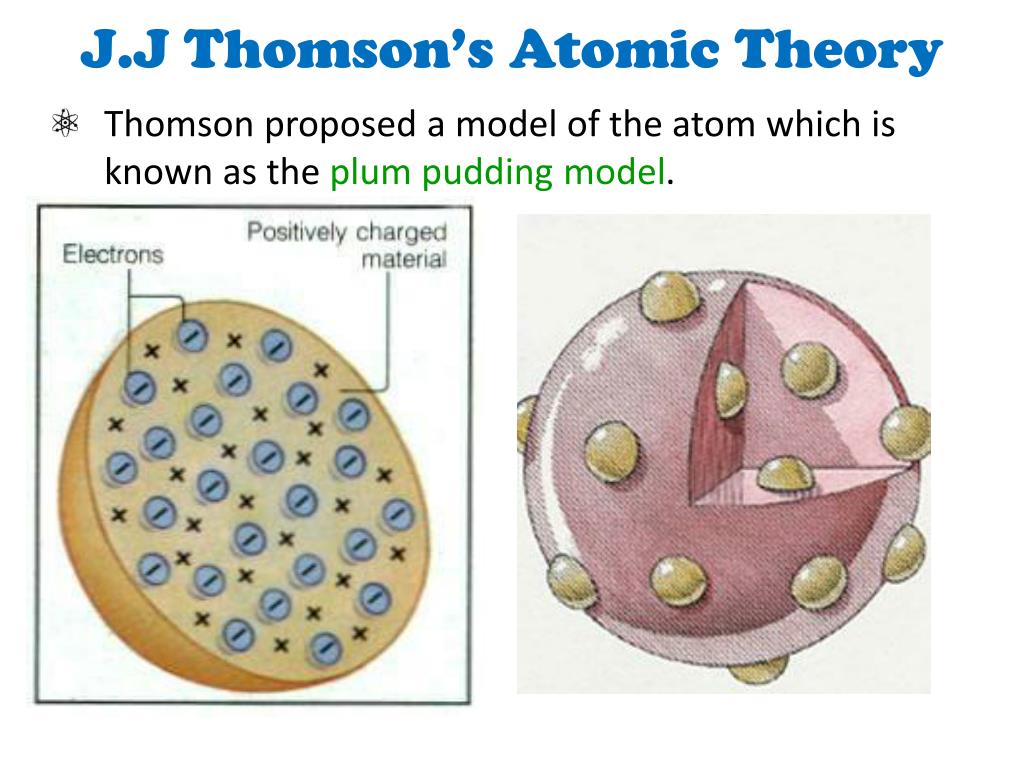
Note: The basic difference between the two models lies in the fact that Dalton proposed that an atom was indivisible and indestructible whereas Thomson worked on the existence of subatomic particles inside an atom and their arrangements i.e., he considered an atom to be a divisible quantity unlike Dalton. The current atomic model, also known as the quantum mechanical model, is a more advanced and accurate representation of the atom compared to Thomsons atomic theory. Atoms of the same element are the same atoms of different elements are different. It consists of three parts: All matter is composed of atoms. The Thomson’ model of an atom did not have any experimental evidence in its support and was therefore rejected. The concept that atoms play a fundamental role in chemistry is formalized by the modern atomic theory, first stated by John Dalton, an English scientist, in 1808. The first evidence-based theory about atoms was given by Dalton in his Dalton’s atomic theory. This model is also known as Thomson’s plum pudding model since it is just like raisins in pudding. > In Thomson’s model of an atom, he proposed that an atom may be regarded as a sphere of approximate radius of $$ cm carrying positive charge due to protons in which negatively charged electrons are embedded or studded. The atom is made up of a number of particles like protons and electrons. Atoms of different elements vary in size, mass, and chemical behavior. Define isotopes and give examples for several elements. Describe the three subatomic particles that compose atoms. Summarize and interpret the results of the experiments of Thomson, Millikan, and Rutherford. All atoms of the same element are identical and have the same mass. By the end of this section, you will be able to: Outline milestones in the development of modern atomic theory. 
The five main points are: Matter is made up of atoms, small and indivisible particles. > However, it was made clear after Thomson's model that atoms are not as simple as suggested by Dalton. Daltons Atomic Theory was formulated by John Dalton in 1808, and it remains a fundamental tenet of chemistry to this day. Dalton did not talk about the internal structure of the atom in his theory. Atoms of the same element are alike in all respects while atoms of different atoms are not the same. > According to Dalton’s atomic theory, the smallest and the ultimate particle of matter is known as an atom. 
Hint: Dalton’s atomic theory proposed that all matter is composed of atoms which are indivisible and indestructible.Thomson’s model of an atom states that an atom comprises electrons distributed between a sea of protons in the same fashion as seeds in a watermelon/plum. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
